|
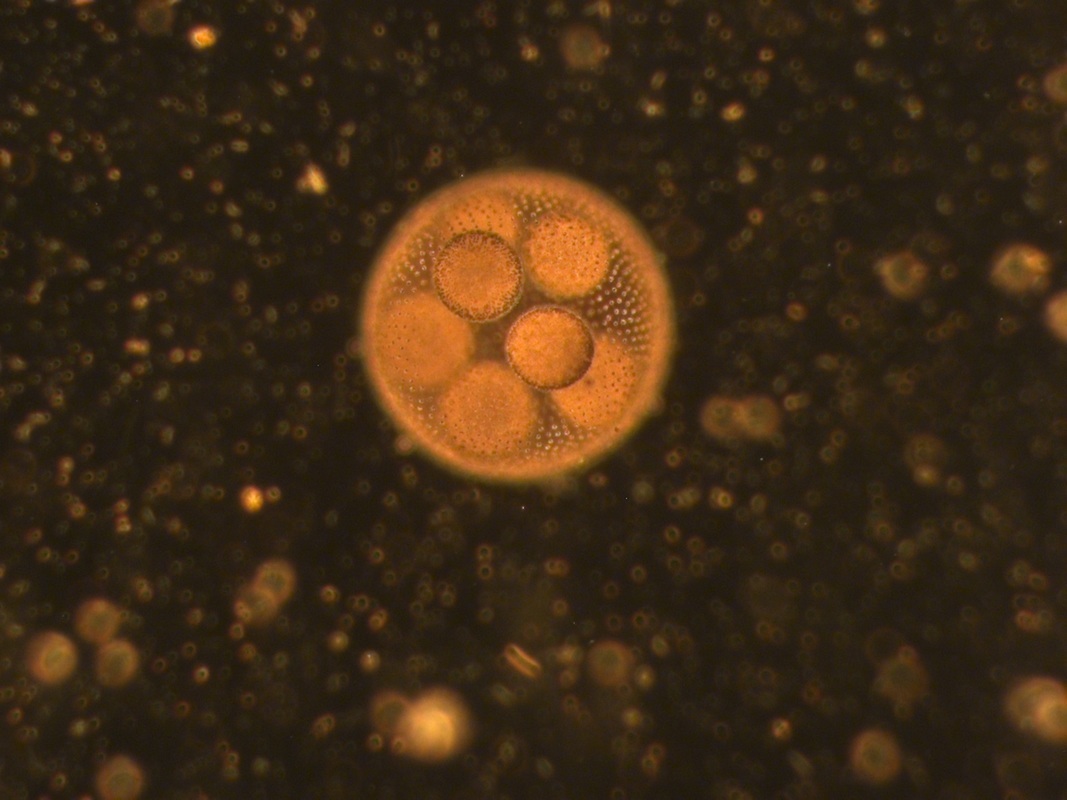
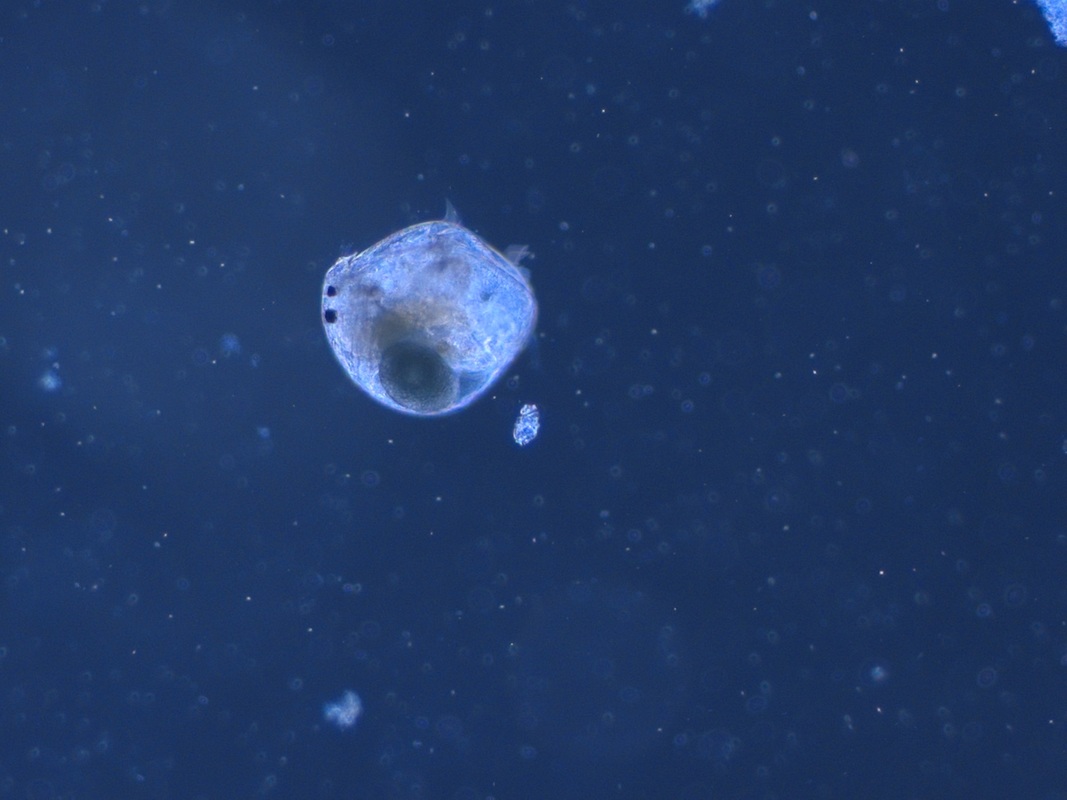
A luxurious volvox A cross eyed bosmina (from David's pond) Some eggs - unknown (to me at least), also from David's pond And last but definitely not least, a daphnia furiously filter feeding.
0 Comments
Last week I took a trip to MARS, not the actual Mars but the Fulda MARS, the where the wolves live MARS and the rather intense world of trying to figure out how aquatic systems (freshwater, groundwater and transitional waters) respond to multiple stressors, from the single water body scale to the European scale. Quite a task, and quite a meeting. I'm involved in two parts of the project, one looking at how cyanobacteria (an indicator of freshwater quality, for ecosystem functioning and for ecosystem services like drinking water and recreation) respond to multiple stressors in single water bodies, using shallow lake experimental tanks - you can hear about this here. The other part is exploring how cyanobacteria respond to the same stressors (climate change and eutrophication) at a Pan-European scale, to do this I am using data from 1300 lakes across Europe to see how the response of cyanobacteria to multiple stressors varies by lake type (depth, size, mixing regime, alkalinity, altitude and so on). Where the wolves live The meeting marked the half way point of the project; during the week we updated each other on our progress and discussed our results so far. The scope of the project is amazing and being delivered by some great minds. What was particularly striking, and something that has impressed me since I started my PhD is the number of women in science, and particularly the number in top positions. I've never really been a pusher of feminism, maybe because I've never felt any oppression in my field, I feel that I have always been encouraged, supported and given equal opportunities. However, it seems fitting to mention this because during the MARS week it was International Women's Day, and on that day I stood up and presented my work as a scientist, was given an opportunity as a woman to be intelligent, to be respected, to use my mind and to be listened to as an equal. It is easy for me to be flippant about feminism on any other day, but only two generations ago none of this would have been possible for me as a woman (just to confirm, I have always been a woman...) and so I think some respectful reflective thanks should be given to the women who paved the way for me to be where I am today. During the meeting it felt quite empowering to be surrounded by an equal mix of men and women who took no care or notice of their sex but instead combined the power of their minds and their individual merits to get on with the job in hand. Because ultimately we are different, in our abilities to communicate, problem solve, conceptualise and organise, and so together we can achieve more. Talking about the shallow lakes experiment - photo credit to the organisers Working on the rivers and lakes experiments data. I won't show you the other one of my screen full of red lines of code, from this angle I can be doing very clever things! - photo credit to the organisers Some of the team of excellent minds! So there you go - women are from MARS too.
Breaking out of my cyanobacteria bubble to think about the bigger picture when it comes to extreme rainfall events. I spent the last week roaming around the lake district fells, climbing the hills along old sheep tracks, battling fierce gales on the top of snow clad mountains and mainly getting wet. Earlier in the week we drove from Coniston over the kirkstone pass ahead of the arrival of storm Frank, anticipating that we may get cut off at Glenridding. A wise, but fortunately for the communities of Cumbria, an unnecessary move – Frank never quite made it. Good job because the river in Keswick was still raging and not far from the flood defences. It was amazing to see the height of the wall plus the glass panels on top of that; to think that this fierce river burst over this barricade is a little bit mind blowing. But then, standing on the top of Blencathra on new years day, you can see the intricate but bare mosaic of troughs and peaks that make up the lake district and from here you can see the torrents of water falling down from the hanging valleys, remnants of the past ice age. What is there on this mainly bare ground to stop the water gushing down? Are we going to continue to build these flood defences higher and higher? In the face of climate change, it looks like we will with storms like Desmond predicted to be 40% more likely. But is fighting a man made problem with a man made solution really the most effective way of battling flood waters?
What is significant about the loss of these upland trees? Tree root growth allows a greater absorption of water, reducing the speed of the flow to the rivers and so provides a natural flood defence against heavy rainfall events. "The rain flashes off sheep pasture as if it were concrete, instantly causing floods downstream. Trees hold back the water and release it gradually, smoothing out the cycle of flood and drought" - George Monbiot, Guardian columnist, environmentalist and supporter of re-wildling. A call then for re-wilding our uplands, back to peatlands and forests? George Monbiot has been campaigning for re-wildling in the UK for some time, in a recent article in UK Hill Walking he answers some key question about re-wildling in light of the recent flood events. In many places attempts to do this have already begun - in the video below some of my former colleagues, Liz Lewis-Reddy and Clive Faulkner from Montgomeryshire wildlife trust talk about the success of a pilot trial of the trust's living landscape project, the Pumlumon project, which aims to improve carbon and water storage by restoring biodiversity in the Welsh uplands. Clive talks of the simplicity behind this idea whilst Liz highlights the socio-economic issues that hinder the wider implementation of the scheme. Reading the views of George Monbiot makes a strong case for re-wilding, but the economic cost benefit in not so easy to campaign as erecting strong, visible barrages. It can be hard too to convince people to change the landscape that we see as being normal but which definitely isn't natural. I'm definitely advocating investing money into conventional flood defences but alongside this there must be also investment in natural solutions that can buffer the response especially in the face of more and more extreme events being predicted. I started identifying and calculating phytoplankton and cyanobacteria biovolumes from the mesocosm experiment. This is great because I love plankton but also not so great because it gives me lots of taxonomic nightmares. I have some great in-house expertise at the Centre for Ecology and Hydrology for phytoplankton and cyanobacteria (Laurence Carvalho and Helen Woods) but sometimes for some trickier ones its good to cast the net a bit wider to get as much advice as possible. As cyanobacteria is now what I'm all about, I need to make sure I get this spot on so I'm hoping the wider cyanobacteria specialist community will give me a helping hand every now and again! General advice: Taxonomic guides: below are the main guides I refer to, those with images e.g. Australian book/ manual of aquatic cyanobacteria and those with drawings and more detailed descriptions - The Freshwater of the British Isles, Manual of aquatic cyanobacteria and the papers by Jiri Komárek on straight and coiled types of Anabaena. I also use the Czech Cyano database. Advice: 1. Suggestions for other useful taxonomic guides? 2. Good formus for discussing taxonomic issues? Specific identification advice: In one of my mesocosms I keep on coming across two different cyanobacteria species which are causing some identification problems (I'm not 100% confident):
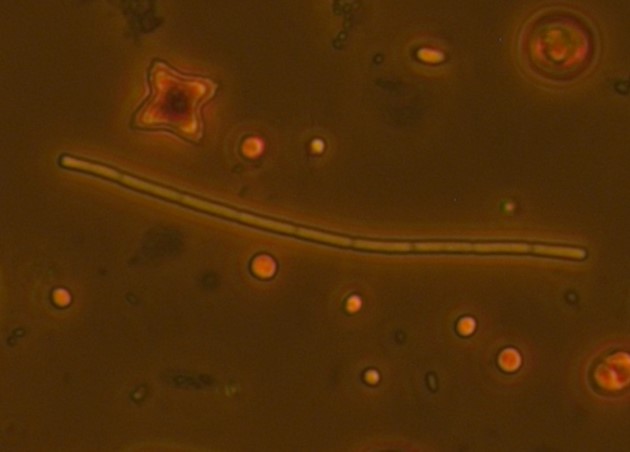
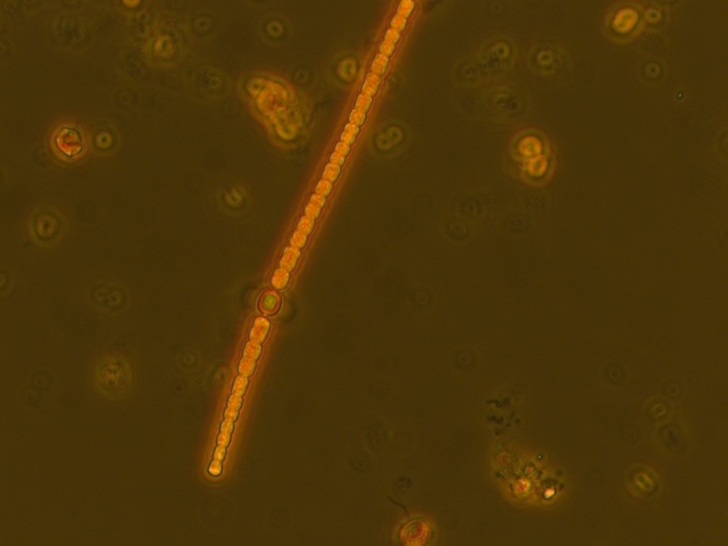
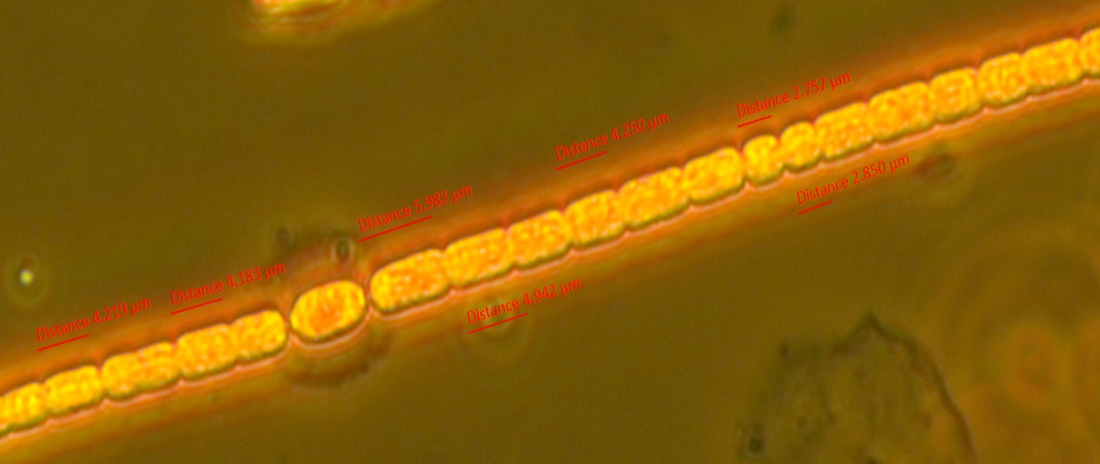
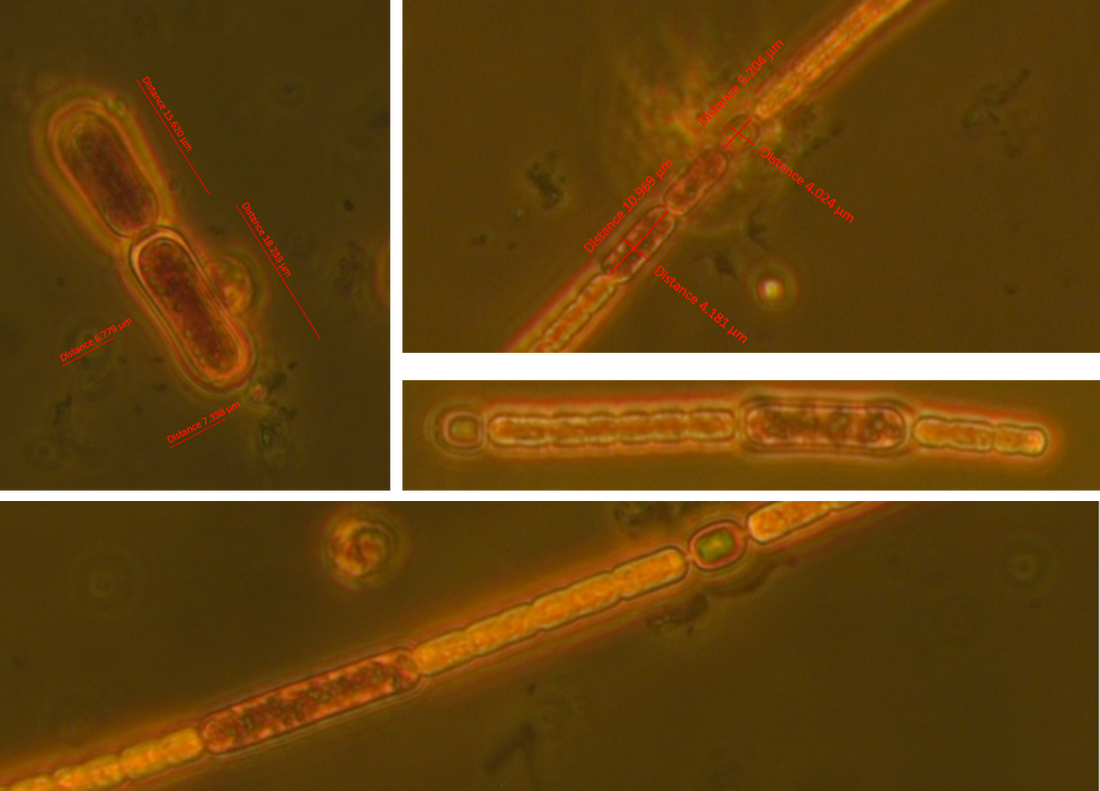
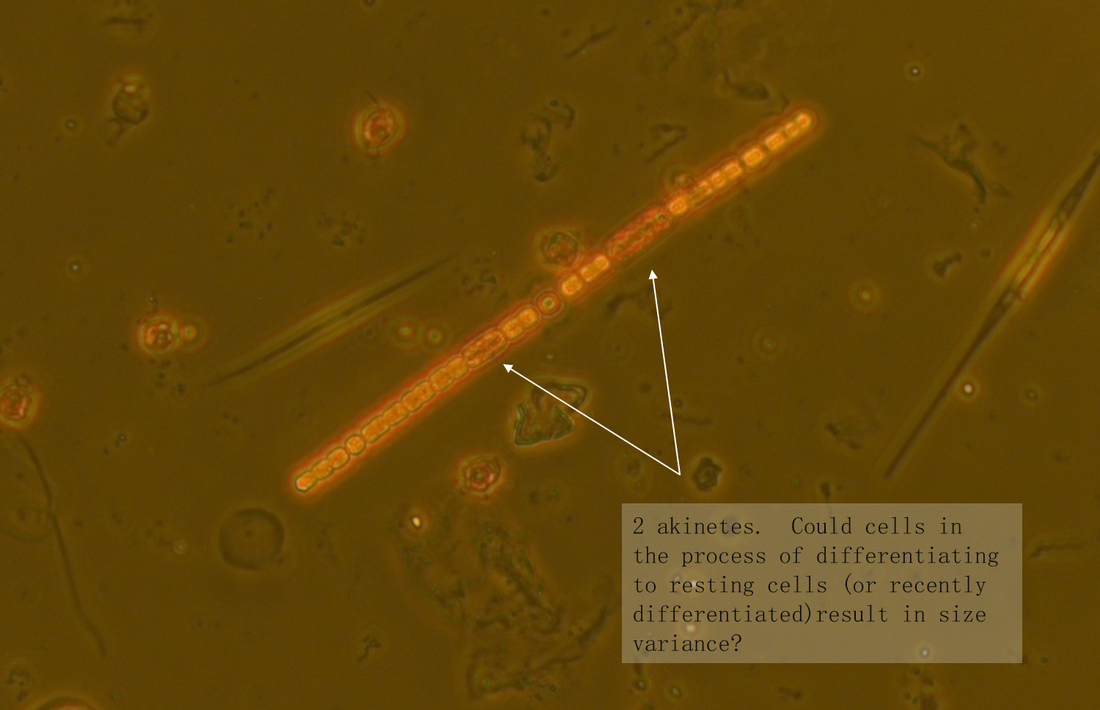
The samples are from heated (+4°C) mesocosms (3000L capacity) in Lancaster (North West England) and are fixed with Lugol's. Images taken in phase contrast at x40 (some at x63) Pseudanabaena vs. Limnothrix: Trichomes solitary and straight, sometimes slightly bent. Trichome average width: 1.5 µm (sample 1 - December), 1.4 µm (sample 2 - June) Cell length (where distinguishable) ~4.4µm In sample 1 the cells were distinguishable with cross walls narrowed and with cells abutting and so were recorded as Pseudanabanea limnetica: Sample 2 - most cells were indistinguishable apart from a few trichomes where some cell walls could be seen so I then questioned whether I was actually seeing Limnothrix redekei rather than Pseudanabaena. The issue: Brian Whitton in - 'The Freshwater Algal Flora of the British Isles' - highlights the taxonomic and nomenclatural problems of Oscillatoria limnetica (Pseudanabaena limnetica) and Oscillatoria redekei (Limnothrix redekei). Specifically he notes that populations of P.limnetica can merely be a growth form of L.redekei whereas it is unlikely that P.limnetica can be a growth form of L.redekei. The advice needed: What should I call this? I would have said Pseudanabaena based on the first sample but the second sample makes me question whether I am just seeing a different growth form of Limnothrix. Or am I actually seeing two different things? I'm suspecting that without genotypic information I won't be able to know with certainty and should note them down as Oscillatoriales however any comments about how other people deal with problematic species like this would be useful. Anabaena vs. Aphanizomenon: Trichomes: Solitary and straight but some bent at heterocyst, cell junction. Ends slightly tapering. Mean diameter at widest point - 3 µm, mean diameter at narrowest point - 2.5 µm. Tapering ends and akinete Cells: See trichome diameter for cell diameters at narrowest and widest points. Length: variable, sometimes hard to distinguish cell boundaries with certainty. From 30 cell measurements: Min: 2.4 µm Mean: 3.7 µm Max: 5.9 µm Heterocysts: Generally longer than wide (4.6 µm x 4 µm ), distant and intercalary. Up to 7 heterocysts per trichome dispersed fairly evenly along the trichome. Some variability in size ± 0.9 µm. Akinetes: Rarer than heterocysts distant and beside heterocysts and singular and seen in a pair. The two seen in a pair were next to a heterocyst. Sizes very variable Akinetes are quite variable in size and shape. Some have slightly dented middles and clearly defined cell walls whilst others are straighter and longer. Unfortunately when I saved the images with the measurements on them in jpeg format the measurements were lost. Size noted down in my note book are: Length µm: 18, 15.6, 19.7, 12 (mean: 16.3 µm) Width µm: 6.7, 7, 4.4, 4.6 (mean: 5.8 µm) The image below shows two akinetes within the same trichome but of differing size. I wonder as akinetes are rare that a potentially nutrient limitation or another environmental pressure has initiated the start of differentiating vegetative cells to resting cells and that I have caught trichomes in the process of doing this? I'm not sure about the natural variability of akinetes within individual trichomes. What I think this is: Aphanizomenon gracile.
Concerns - instinctvely I thought it was Aphanizomenon however I was unsure about the degree of tapering at the end. Looking through all the keys cell size appears to be to big for most Anabaena species but I want to double check. Advice: correct identification - please! Yesterday I was down in Windermere for the day doing the last few chlorophyll a extractions for the mesocosm experiment at the FBA - we still have a lab there from back in the days when the centre for ecology and hydrology was located there, the good old days I bet. Lake, mountains, science and plankton - my favourite things. While I was making a cup of tea, I was pounced on by Gary Rushworth who took the opportunity to tell me about the 'Clear Waters' oral history project, which he was the project officer for a few years ago. And now I'm going to tell you about it, because I think it's pretty cool. The Clear Waters oral history project tells the story of lake Windermere over the past 50-60 years, through the memories of the people who have lived and worked there. I love this kind of history, based on memories, old photographs and stories; tales of floating hen houses, monster fish and ice skating on the lake. The project interviewed volunteers from four different groups of people: anglers and sport fishermen, local people involved in lake recreation, long term residents with an interest in wildlife or recreation and former employees and members of the Freshwater Biological Association. The project exhibited in and around Windermere but some extracts and information can be found on the project website. Comparing sizes - I bet she swapped the big fish she caught with his ... well done old chap you really outdid me there. One for the #distractinglysexy album It's interesting to hear people talk about their experiences of the lake but it also documents how FBA scientists and local residents have viewed the changes seen in the Windermere catchment since the Second World War, changes such as the loss of reedbeds and natural shore line, increases in boat moorings and changes related to climate such as reduced ice cover during the winter. These views, formed by their individual experiences of the lake help us understand the complex relationship between lakes and human activities. Understanding and quantifying the relationships between people and ecosystem services is an important step in making the most effective management decisions. Go check it out! Oi, cyanobacteria! Have you got anything to say about this ...
I'm hoping that the inclusion of 'extreme' in the title will get you hooked... This is back dated, it actually happened at the start of August but I have been super busy with other field work, courses and writing literature reviews. The aim of this mesocosm experiment is to explore the response of lake ecosystems to multiple climate change and nutrient stressors. One of the effects of climate warming is a change in the water cycle, it is predicted that northern European countries will have more rainfall annually with an increase in rainfall during the winter, overall summers will be drier but with an increase in extreme rainfall events lasting 1-5 days during the later summer (July to September) whilst southern European countries will experience a reduction in annual rainfall (Christensen and Christensen 2003). Modelled changes in precipitation patterns will result in 10-30% decreases in run-off in southern European countries whereas northern countries will experience a 10-30% increase in run-off (Milly et al 2010). So as well as a 'warming' treatment we also have an 'extreme rainfall event' treatment. Water (1500 litres) is pumped into each tank to mimic the increase in in-flow that will happen as a result of a high intensity rainfall event. The tanks hold 3000 litres so this is similar to displacing half of the water in the tanks, although this is not direct displacement as just like in a lake the water will mix with the inflowing water and so the water lost from the tank will be a mix of the water from the mesocosms and the water pumped in. This event happens once every 12 weeks, 4 events in total over the duration of the experiment. Preparation: For each extreme rainfall event we need 24,000 litres of water - at the top of the site we have two large tanks which we fill up with filtered water from a nearby reservoir (this does not contain algae but does contain nutrients which we adjust for at the next nutrient addition). The tank is open to the elements so after 12 weeks of being exposed to rainfall, leaves and what not the tanks need to be cleaned out until they are spotless. This involves lots of buckets, a step ladder and a pair of waterproof overalls. The finished product, tank spotless and ready to be filled with water. If there were ever a picture of an empty tank that I would like to sit and stare at for a long time it would be this one, oh the satisfaction! Now the taps get switched on, it takes 24 hours to fill both up so we leave them running overnight ready for the event the next day. The rainfall event: We place one pump in each large tank and attach a long hose pipe that will reach to all of our smaller experimental tanks. Each pump has a different flow rate so first we time how long it takes to fill up 50 litres in marked buckets and multiply this to work out the time needed to pump 1500 litres into each tank for the different pumps. Then we place the hose end in a bucket so that the force of the water hits the bottom of the bucket instead of churning up the sediment and switch on the pumps. Easy peasy, no stress involved at all! It's like a rocket launch for each tank - five, four, three, two, one, GO - but probably less exciting. The end of the video below shows a short clip of a tank being treated with this treatment, to get an idea of the magnitude of the event the duration of the treatment is 15 minutes for one pump and 21 minutes for the other pump. At the start of the video you will see me using the algaetorch to measure chlorophyll a and cyanobacteria biomass in the tanks. I do for all tanks but twice for the rainfall treated tanks, once before the treatment and once afterwards. We also do all the usual in-situ measurements as described here. A tank mid treatment - now the cover photo for the blog will hopefully make sense! The day after the rainfall event we take water samples and process them as described here . Usually we sample every four weeks but when we do a rainfall event we sample the day after, a week after and three weeks after (which coincides with the usual sampling timetable). So now that you know what's involved, lets go back to why are we interested in the response of aquatic ecosystems to changes in flow. Increased flow through a lake results in losses of algae biomass from the direct effects of flushing (algal biomass is lost in the flow) e.g. Carvalho et al (2012). Previous research has demonstrated that phytoplankton community composition is strongly modulated by the amount, duration and seasonality of flow (different taxonomic groups are more sensitive to flow than others); cyanobacteria are a slow growing taxa and prefers slow, stable and warm conditions whilst other faster growing phytoplankton like diatoms do well in fast flowing, mixed environments (e.g. Reynolds et al (2012)). We are interested in how water bodies will respond to these periodic episodes of high flow where biomass will be flushed out of the system and the effect it will have on cyanobacteria in combination with other stressors - warming and nutrient enrichment. How long does it take for the biomass to recover to its initial state? Is the composition different before and after flooding? Is the response different in tanks that are also treated with warming, excess nutrients or both? and so on. One last thing - for the past 3 months Nur Filiz a PhD student from METU has been helping out with the mesocosm experiment. Nur is also involved in the MARS project and is looking at the 'Impact of Climate Induced Extreme Events on Phytoplankton Community in Shallow Lakes'. During her time here she has been doing some phytoplankton counts from previous sampling occasions and has been helping me with the running of the experiment. This was Nur's last day - thanks for your help Nur!
Carvalho, L., C. Miller, et al. (2012). "Water quality of Loch Leven: responses to enrichment, restoration and climate change." Hydrobiologia 681(1): 35-47. Christensen, J. H. and O. B. Christensen (2003). "Climate modelling: Severe summertime flooding in Europe." Nature 421(6925): 805-806. Milly, P. C. D., K. A. Dunne, et al. (2005). "Global pattern of trends in streamflow and water availability in a changing climate." Nature 438(7066): 347-350. Reynolds, C. S., S. C. Maberly, et al. (2012). "Forty years of monitoring water quality in Grasmere (English Lake District): separating the effects of enrichment by treated sewage and hydraulic flushing on phytoplankton ecology." Freshwater Biology 57(2): 384-399. ... sleep deprivation and a wet head. Night zooplankton sampling one of twenty seven. Actually an extension of day two because of some really bad weather coming in. After dark we take a 5 litre integrated sample from each tank. We do this at night to make sure of an equal distribution of zooplankton within the mesocosms - during the day they will hide in the macrophytes from their fish predators. The water is then filtered through a 20µm mesh and then the zooplankton caught are washed into a bottle before being preserved with ethanol. Identification and counts (the most fun part) happens at a later date.
How wrong I could be. At the height of summer sampling start times were as late as 11.30 p.m, and end times as late as 2.35 a.m because of course now that it is summer its light for longer and more of the tanks (all of them it seems) have high algal biomass so trying to filter five litres of water per tank through a 20μm gauze can take some time. I have never been so happy for the days to get shorter again. Sometimes when algal biomass is really high its just not possible to get the sample through a 20μm gauze so we filter through a 50μm instead. This still captures most of the zooplankton but the mesh size is too big for some of the smallest rotifers. During the winter all of the tanks would go through a 20μm gauze without any problems, below shows you the situation at the end of August - mainly 50μm filters. It demonstrates nicely how different the biomass is between the winter and summer months. This last ever night zooplankton sampling finished at 10:25pm, all went well, a bit windy but no rain, until I was packing up the buckets and out of nowhere one big torrential downpour hit me. From bone dry to soaked in a matter of seconds. Boy am I glad that its all over!
All in the name of science. Plankton will make me do the strangest things. ... a very long day and almost the death of my filtering pumps.
I then drive the samples back down to the lab, pop them in the fridge and get ready to filter for the next four or five hours. First I preserve 100ml of unfiltered water from each tank with Lugols, this is for algae identification and bio-volume counts. I send off 50ml unfiltered water from each tank to chemistry for total nutrient analysis (the nutrients found in the water and also within organisms). The rest of the water sample gets filtered like you can see below. I need one filter for chlorophyll a analysis (we do laboratory analysis as well as using the sonde ) and one filter for cyanobacteria cellular toxin analysis. Toxins can be within cells or released into the water so I also need to take a certain volume of the filtered water for in-water toxin analysis. Some of the tanks are so high in biomass that obtaining the right volume of water for toxin analysis takes a long time and a lot of filters. Below is pump one of two, they run continuously. I started filtering at 10am and finished at 3.15pm - the poor pumps felt a bit hot by the end. The remainder of the filtered sample is split into another sample for nitrate analysis which the chemists process and the other part for srp (soluble reactive phosphorus) analysis which I will do later in the afternoon. Nutrients can be held within organisms or are available in the water column, total nutrients is a measure of both whilst srp calculates what is available in the water. Cells will release these nutrients when they die and so a high srp in a closed system like the mesocosms can be indicative of a break down of biomass. Once the filtering is done I get on with srp analysis. This involves chemistry, reactions, sulphuric acid which smells pretty bad, wearing safety goggles which make me look very attractive and a spectrophotometer, which I can't say properly.
And that marks the end of Wednesday, well usually it would. Because of the weather day three, Thursday is being moved to day two so I have to now bike home to get the head torches (which I forgot) to start on part three, night zooplankton sampling. ... lots of data and some dead fish. This is actually the last sampling event (one of twenty seven) for the mesocosm experiment. So in case I miss it here is a break down of what happens when we go and sample. Day 1: in situ data collection (sondes, algaetorch and nephlometer), periphyton and maintenance Day 2: collection of water samples, filtration in the lab for toxins and chlorophyll a, srp analysis and sample fixation for bio-volume counts Day 3: Night time zooplankton sampling Day 1 - Tuesday, in situ data collection As soon as I get to the mesocosms the Exo2-sonde goes into the first tank, we take averages from at least six readings which takes four minutes per tank, four times thirty two equals lots of minutes so I get that started straight away. The EXO2 is a muti-parameter sonde that measures temperature, conductivity, salinity, pH, dissolved oxygen, chlorophyll and blue-green alage (cyanos). Measurements are logged internally which means I can get on with taking some other readings. The algae torch also measures chlorophyll a and cyanobacteria biomass using fluorescence of algae cells and is used to get an in-situ measurements. I, as a plankton geek think it pretty cool! The nephelometer measures turbidity (suspended particulates) by recording the light reflected from the particles (from a beam of light emitted from the instrument). As I go around I also check all the tanks for any maintenance issues and clean all the sensors (temperature, oxygen and PAR - photosynthetically active radiation) located in the tanks. I also make any observations of 'unusual' activity in the tanks, like these dead fish found below. I'm not entirely sure why there were so many dead right now, the only thing immediately different about this tank is the amount of dissolved oxygen (%) - 27% compared to >65% in the rest of the tanks, which could be a factor.
To account for this, periphyton growth is measured on a monthly basis in each tank by calculating the dried weight from pre-measured in-situ strips. The picture below shows the difference in the bio-volume and type of periphyton growing in each tank which demonstrated nicely the need to account for this effect. And that marks the end of the Tuesday session up at the mesocosms. All I need to do now is enter the data and prepare for the sampling marathon tomorrow.
Over the past few weeks Euglena sanguinea has taken a bit of a liking to our mesocosms ... Aspect number one: What is that? "Hey (generic alien/scary film character name), come and look at this...." Aspect number two: "Don't touch that s***, it moves!" It makes you want to double up on gloves!  These birds are the unfortunate casualties of this horror story- don't worry the main character mysteriously survived and is now casually sipping a Martini in the lab. The surface scum is very densely packed and moves almost as a mass ; its quite hard to describe but its a bit like the skin that forms on custard (but less appetising) or probably more in line, the mouldy skin that forms on a cup of tea when you have left it on your desk for too long (I assure you that I have never done this). I should stress that not all of the tanks are so 'bad' (actually only two, as far as I have observed) and in the one featured above a fight for supremacy has been going on between Euglena sanguinea and Anabaena spirulina. On a serious note - are there any trends to these blooms? On the surface of it all not really - both of the tanks with the dense scums are nutrient enriched tanks, but one is heated and the other is unheated. Many of the tanks have some reddish tinge to them (confirmed to be E.sanguinea after a peak under the microscope) with varying degrees of scum development; they look more like the 'after' photo below. So, it could be a case of different responses between tanks because of different exposure to mixing from the wind. The clear ability for E.sanguinea to form dense surface blooms means that it can compete with other buoyant species like Anabaena and Microcosytis; seeing as my analysis will be investigating the response of cyanobacteria to different stressors this is definitely one to keep my eye on. What a difference a windy weekend makes ...
|
AuthorI have managed to turn my rather unhealthy obsession with plankton in to my day job. Things don't get much better than this! This blog documents my PhD research and the plankton delights I encounter along the way. Archives
June 2016
Categories |


































 RSS Feed
RSS Feed
